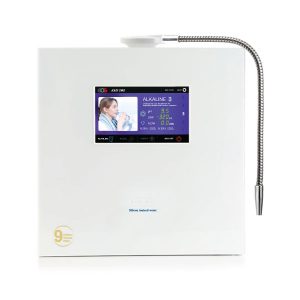
performance on your health. Thanks to its
new SMPS electronic transformer.





16 layers filtration
than the REVELATION II, but with a whole new
designer faucet. Add style to your decor.




Shipping
Québec, Canada, USA
Free shipping
For water ionizers
Free Formation
To use your product
24H/7J Support
Ask us your questions
Alkaline ionized water products designed for your reality!

For over 10 years, Aqualife has brought you the benefits of alkaline ionized water with a wide selection of ionizers, filters and pH testers.
- Free delivery, even on weekends
- Cheapest prices on the market
- Same day delivery
- Telephone service available 7 days a week
- Advisory service on the use of your products

RIO – under sink alkaline water ionizer (pH 11, 9 plates, double 16 layers filtration) – EOS Hitech
*** FREE SHIPPING FOR CANADA AND THE USA ***
The EOS Hitech RIO under sink water ionizer combines discretion, design and adaptability. The perfect model to enjoy the double filtered and invigorating alkaline ionized water.
TUV Certification (High End product)
Thanks to its high quality 9 titanium plates, the RIO guarantees you at all times a water rich in molecular hydrogen (from 1200 to 1450 ppb) and above all, free of any toxicity and unwanted residues. In addition, with its 16 layers double filtration, you can count on water rich in negative ions. And we say "negatives ions" we say "beneficial health effects"!
This exceptional model also contains an SMPS electrical transformer, which efficiently manages electricity consumption during the water ionization process.
NEC 901 / JAY 201 – alkaline water ionizer (pH 11, 9 titanium plates) – EOS Hitech
*** FREE SHIPPING FOR CANADA AND THE USA ***
The NEC 901 / JAY 201 countertop water ionizer, manufactured by EOS hitech, produces alkaline ionized water rich in molecular hydrogen (1200 to 1450 ppb) and above all, highly revitalizing.
TUV Certification (High End product)

The NEC 901/JAY 101's double 16 layers of filtration provides additional minerals to your water, in ion form. Consider calcium and magnesium, which are full of health and well-being benefits.
And what about its design ,that makes it a sleek high-end model, which can easily match all countertops!
In addition, this counter water ionizer can be easily installed in less than 10 minutes without the need for the plumber or you can plug it directly to your cold tap water under your sink with a diverter (not included).
Learn more about this exceptional ionizer, which will meet many of your health goals.
Bawell BW-6000 alkaline water ionizer, 7 plates, double filtration (model 2195)
- 4 different alkaline levels (to meet different needs)
- 3 acidic levels
- 1 pure water level
- 29 different settings for water that suits you 100%
- Digital and touch screen, LED light
- Water flow control
REVELATION II – under sink water ionizer (pH 11, 9 plates, double 16 layers filtration) – EOS Hitech
FREE SHIPPING FOR CANADA AND THE USA.
The EOS Hitech REVELATION II under sink water ionizer combines discretion, design and adaptability. The perfect model to enjoy the double filtered and invigorating alkaline ionized water.
Thanks to its high quality 9 titanium plates, the REVELATION II guarantees you at all times a water rich in molecular hydrogen (from 1200 to 1450 ppb) and above all, free of any toxicity and unwanted residues. In addition, with its 16 layers double filtration, you can count on water rich in negative ions. And we say "negatives ions" we say "beneficial health effects"!
This exceptional model also contains an SMPS electrical transformer, which efficiently manages electricity consumption during the water ionization process.
Take advantage of this high end alkaline water ionizer to achieve your health goals.
GENESIS 1 – counter top water ionizer (pH 10, 5 titanium plates) – EOS Hitech
*** FREE SHIPPING FOR CANADA AND THE USA. ***
The Korean company EOS Hitech is a global reference for water ionizers. Just try one of their products to understand why.
TUV Certification ( High End Product)
Among the EOS Hitech ionizers available is the Genesis 1: a countertop water ionizer with full of health and wellness benefits. Choosing this model means opting for the high end at a low price!
5 titanium plates, covered with platinum, as well as 2 active charcoal filters, give Genesis1 the ability to effectively filter waste and toxins, such as chlorine, from your water. This can reach a pH of 10, in addition of large level of molecular hydrogen (from 900 to 1100 ppb).
To top it all off, the Genesis 1 water ionizer installs in less than 10 minutes on any standard faucet. Not to mention its high-performance electrical transformer, which ensures you good electricity management.
And with more than 15 certifications, you can be assured of a reliable, stable and efficient ionizer. Take it everywhere to enjoy pure, invigorating and fully alkaline water at all times.
Bawell alkaline water ionizer pH 10, 5 plates, BW-SM1
The handy and convenient Bawell Alkaline Water Ionizer installs in less than 10 minutes on a standard faucet.
Functional design and superior technology.
The Bawell Water Ionizer is state-of-the-art and combines ease of use, durability and beauty. It helps to help improve your health in ways that you never thought possible to be possible ....
US and International Sales Only
LYDIA 8080 – Counter top alkaline water ionizer pH 10+, 5 plates
Installs in less than 10 minutes on a standard kitchen faucet without a plumber.Modern and Zen design. TUV certification, superior quality. Ionizer high-end.
Portable alkaline filter AOK-909
- Converts ordinary water into delicious, healthy water
- Impregnates drinking water with antioxidants, energy, minerals and other essential elements of water filters with 8 layers of filtration
- Eliminates 99.99% of the bacteria in tap water
- Removes dirt, limescale, pesticides, chlorine, smell of chlorine, fluorine, mercury, lead, copper, iron, aluminum, and other harmful chemicals, heavy metals and pollutants ordinary water
- Softens ordinary tap water
- Moisturizes the body by improving absorption
DISCOVER THE POWER OF ALKALINE IONIZED WATER

Tap water is often oxidized and contain toxins and other undesirable materials, such as chlorine or pesticides. Nothing to help you achieve your health goals!
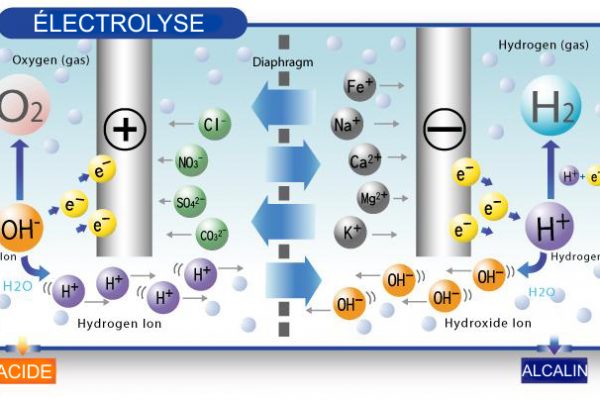
Fortunately, Aqualife offers water ionizers that, thanks to filters, negative ions and a process called electrolysis, add many benefits to the water you drink every day:
- It becomes more alkaline (anti-acid)
- It slows down the oxidation (premature aging) of your cells
- It is enriched with hydrogen and good minerals
- It promotes the right balance of your body
- It rehydrates the body and eliminates waste
Installed on countertops or under a sink, these ionizers are a jewel of simplicity, performance, design and durability. They adjust the pH of the water according to your needs. Alkaline ionized water produced by Aqualife ionizers is suitable for all profiles and ages.
Say ``NO`` to water bottles!
Each year, more than 400,000 barrels of oil are used to make plastic from water bottles. And only 30% of these will be recycled!
Water ionizers (Countertop, under sink & portable) reduce this waste of resources, which creates irreparable damage to the environment.
1 million plastic bottles are bought every minute around the world. Each bottle pollutes our planet a little more. Let’s get into the habit of always having a reusable water bottle and filling it with an alkaline water ionizer.
A water ionizer is cheaper than buying bottles of water for much better quality water than bottled water.
Think green with Aqualife water ionizers.
SOLUTIONS ADAPTED TO YOUR NEEDS
DISCOVER OUR PRODUCTS
WATER IONIZERS
WATER FILTERS
LOOKING FOR WATER TESTERS OR INSPIRING BOOKS?
GET INFORMED!
ALL ABOUT IONIZED WATER ALKALINE
Learn more about the benefits of water, alkaline ionized water and hydrogenated water.
WHAT IS ORP ?
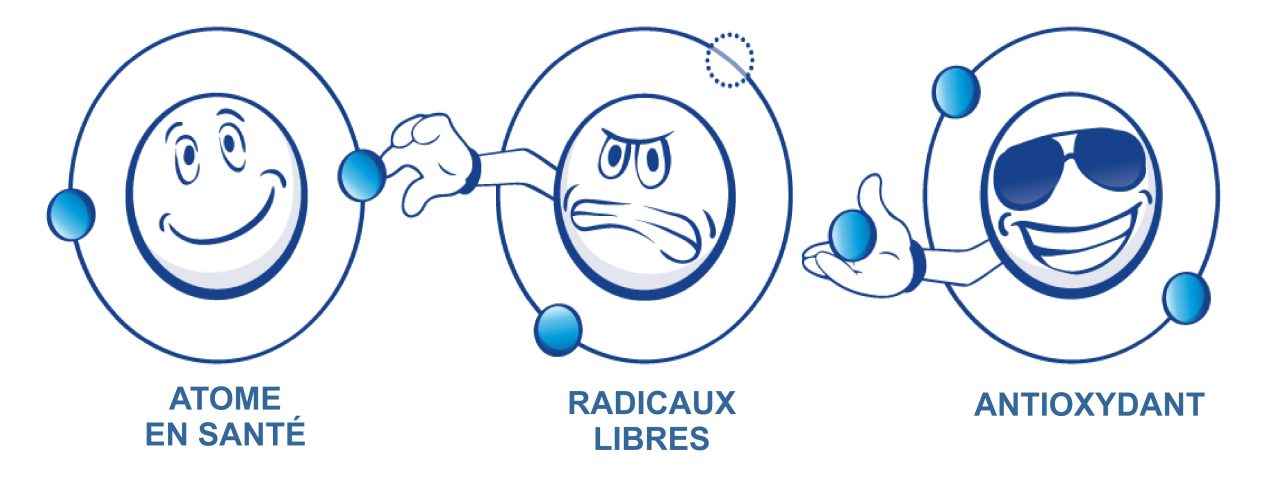
The oxidative potential of water determines its ability to reduce oxidation and thus prevent premature aging of your body. The lower the ORP, the greater the reduction potential …
WHAT IS THE pH ?
Hydrogen potential (pH) is a unit of measurement that determines the level of acidity or alkalinity (basicity) of a solution. The higher it is, the more alkaline the liquid. However, there are levels to avoid …
pH CHART
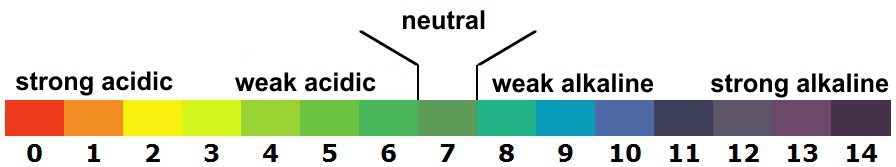
A scale that allows you to accurately measure the level of acidity or alkalinity of the water, as well as the effects on your health. Discover the ideal pH levels for your alkaline ionized water:
- pH of 8.5: the ideal level for alkaline ionized water drinkers who want to familiarize themselves with it.
- pH of 9: an optimal pH level, which allows you to drink alkaline ionized water unlimitedly.
- pH of 9.5: This pH level increases your energy and vitality level, as well as reducing your body’s recovery time after exercise.
- pH of 10: a higher alkaline level, which can be used for cooking or for making tea/coffee.
- pH of 11: a pH level that is not designed to be drunk by humans. By case, it can be used to clean or remove pesticides on your fruits and vegetables.
LIVE BETTER, EVERY THROAT!
AQUALIFE
At Aqualife, we are dedicated to health, both ours and yours. That’s why we distribute high end alkaline ionized water products around the world that make a difference in the lives of thousands of people.
VHere are the foundations and values that drive us every day:
- With good lifestyle habits, we believe your body can heal itself.
- With good health information, you can make informed choices that improve your quality of life.
- With good products, we can prevent and curb many health problems.
- With affordable prices for these products, anyone can discover the health benefits of alkaline ionized water and enjoy a healthy lifestyle.
Welcome to the first step in your journey to optimal health.

LEARN MORE ABOUT THIS?
NEWSLETTER
Stay informed of our latest promotions.
OR FOLLOW US
NOTICE
Aqualife has not verified the accuracy of the information in the documents and the website. Please note that Aqualife has not conducted a specific scientific study for the facts set out on documentations and website. The information on the site and documents are taken from the web. You need to do your own research on the properties of ionized alkaline water. All information on this website is for reference and for academic purposes only.
**** Purified water (alkaline ionized or acidic) is not a treatment for a medical condition. If you have a medical problem, please consult a health care professional for specialized medical advice. Alkaline wateris simply purified and restructured from tap water (without chemicals or acid molecules). It does not contain any drug substances to cure your illnesses. Ionized water, such as fresh air or good food, helps your general health, but is not a substitute for medical treatment. See your doctor if you have any doubt.
Have questions about Aqualife or our products?
Get in touch!